Cardiovascular diseases and cancer are, individually and collectively, the leading causes of morbidity and mortality worldwide, sharing similar epidemiological characteristics and risk factors, such as population aging, comorbidities, lifestyle habits, environmental exposures, and sociodemographic determinants (1,2). In this context, the intersection between cardiology and oncology is no longer circumstantial and has become a defining pillar of contemporary medical practice.
In the last decade, the introduction of cancer immunotherapy has substantially modified the natural history of neoplasms previously associated with extremely poor prognoses. Tumors such as metastatic melanoma, lung cancer, renal cell carcinoma, and other advanced neoplasms have shown significant survival gains after the incorporation of immune checkpoint inhibitors (ICIs), among other strategies (3,4,5). This is a concrete change in the natural history of these diseases, with direct implications for the organization of health care (3).
This therapeutic revolution has brought with it a new clinical reality. A subset of patients currently receiving immunotherapy or with recent exposure to it have begun to present cardiovascular manifestations related to immune-mediated toxicity, with a heterogeneous spectrum ranging from subclinical changes to severe, potentially life-threatening conditions (2,3). These manifestations may require anything from lifestyle changes and specialized clinical follow-up to complex pharmacotherapy, percutaneous interventions, and, in selected cases, highly complex cardiovascular surgical procedures, such as heart transplantation (6).
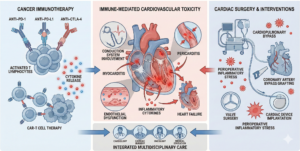
The interface between cancer, immunotherapy, and cardiac surgery thus inaugurates a field that is still undergoing consolidation (Fig. 1). This evolving domain requires an in-depth understanding of the pathophysiological mechanisms of immune-mediated toxicity, refined preoperative assessment, and close attention to perioperative complications.
ICIs act by removing physiological mechanisms of immune self-regulation, allowing T lymphocytes to recognize and eliminate tumor cells more effectively. However, this systemic immune activation can result in loss of self-tolerance and aggression against healthy tissues, including the myocardium, conduction system, pericardium, and vascular endothelium. ICI-associated immune toxicity can affect virtually any organ and presents with a broad spectrum of clinical manifestations, from mild to fulminant forms (3,5).
Among the cardiovascular manifestations, immune-mediated myocarditis stands out as a relatively rare event; however, it is associated with high mortality, especially when it occurs early after initiation of therapy or is accompanied by complex arrhythmias and atrioventricular block (7). In addition, heart failure, subclinical systolic dysfunction detected by reduced global longitudinal strain, pericarditis, pericardial effusion, and acute ischemic events have also been described in the literature.
From an epidemiological standpoint, ICI-associated myocarditis, although considered rare, occurs in approximately 0.04% to 1.14% of treated patients, but carries a high case-fatality rate, ranging from 25% to 50%, substantially exceeding that observed with many other immune-related adverse events (8,9). These data underscore that, despite its low incidence, this complication has significant clinical impact.
Cell therapies, particularly those involving genetically modified T lymphocytes, such as chimeric antigen receptor (CAR)-T cells, have a distinct profile of cardiovascular involvement, predominantly mediated by cytokine release syndrome. This systemic inflammatory state is characterized by intense vasodilation, hypotension, transient myocardial depression, arrhythmias, and distributive shock, often requiring advanced hemodynamic support. Although many of these changes are reversible, their occurrence has important implications for surgical risk and perioperative management (9).
With the progressive expansion of immunotherapy, a statistical increase in the number of cancer patients who, at some point during follow-up, will require invasive cardiovascular procedures, such as myocardial revascularization, valve repair, correction of structural complications, or cardiac device implantation, is inevitable (1). In some of these cases, cardiac pathology is directly or indirectly related to treatment-related toxicity, which poses additional challenges to surgical planning and clinical management.
In the setting of CAR-T cell therapy, cardiovascular complications have been reported in up to 26% of patients, primarily driven by cytokine release syndrome, which represents the main underlying pathophysiological mechanism. In severe cases, these complications may progress to acute heart failure, complex arrhythmias, and cardiogenic shock (4,5).
In the context of cardiac surgery, these particularities take on even greater relevance. Cardiopulmonary bypass triggers a significant systemic inflammatory response, and in patients previously exposed to immunotherapy, this reaction may be exacerbated, increasing the risk of hemodynamic instability, acute ventricular dysfunction, and perioperative arrhythmias. A history of cancer alone is no longer sufficient for adequate risk assessment; understanding the type of immunotherapy used and its immunological repercussions is essential (4).
The preoperative evaluation of these patients should be comprehensive and individualized, incorporating a detailed characterization of the immunotherapy administered, the interval since the last dose, the presence of previous immune-mediated adverse events, and systematic evaluation using cardiac biomarkers and advanced imaging methods. The integration of cardiology, oncology, and cardiac surgery is not merely a theoretical recommendation, but an evidence-based practical necessity.
References
- Alhuneafat L, Guha A, Blaes A, Konety SH. Cancer and cardiovascular disease: shared risk factors, mechanisms, and clinical implications. JACC CardioOncol. 2025;7(5):453-69.
- Lyon AR, López-Fernández T, Couch LS, Asteggiano R, Aznar MC, Bergler-Klein J, et al. 2022 ESC Guidelines on cardio-oncology developed in collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS). Eur Heart J. 2022;43(41):4229-361.
- Palaskas N, Lopez-Mattei J, Durand JB, Iliescu C, Deswal A. Immune checkpoint inhibitor myocarditis: pathophysiology, diagnosis, and management. J Am Heart Assoc. 2020;9(2):e013757.
- Du H, Wang J, Wang Z. Cardiovascular adverse effects of immunotherapy in cancer: insights and implications. Front Oncol. 2025;15:1601808.
- Postow MA, Sidlow R, Heller G. Immune-related adverse events associated with immune checkpoint blockade. N Engl J Med. 2018;378:158-68.
- Issa AFC, Guimarães TCF, Vidotti V, Zagni G, Santos M, Miranda J. Transplante cardíaco em pacientes com cardiotoxicidade por quimioterápico. ABC Heart Fail Cardiomyop. 2022;2(4):425-7.
- Mahmood SS, Fradley MG, Cohen JV, Nohria A, Reynolds KL, Heinzerling LM, et al. Myocarditis in patients treated with immune checkpoint inhibitors. J Am Coll Cardiol. 2018;71(16):1755-64.
- Zhou F, Liu G, Zhang S, Luo C, Hu S, Wan S, et al. Cardiotoxicity in cancer immunotherapy: a systematic review and global meta-analysis. J Transl Med. 2025;23:718.
- Neelapu SS, Tummala S, Kebriaei P, Wierda W, Gutierrez C, Locke FL, et al. Chimeric antigen receptor T-cell therapy: toxicities and management. N Engl J Med. 2018;378:439-51.